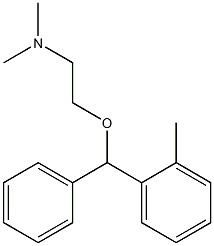
What Makes Certain Formulations More Resistant to Oxidation and Degradation?
2026-03-02
Why Does Oxidation Occur in Modern Chemical and Pharmaceutical Systems?
Oxidation is one of the primary pathways that compromise stability in active ingredients, intermediates, and finished dosage systems. In pharmaceutical and specialty chemical manufacturing, oxidative stress may be triggered by exposure to oxygen, trace metals, light, elevated temperature, or residual moisture. These factors accelerate free radical formation, initiate chain reactions, and eventually lead to potency loss, discoloration, viscosity shift, or off-odor development.
In regulated markets such as the United States and Europe, stability performance is evaluated under ICH climatic conditions, including long-term and accelerated testing. Products that fail oxidative stability requirements often experience reduced shelf life, increased recall risk, and regulatory delays. Our factory has observed that improper excipient compatibility and insufficient antioxidant systems remain two of the most common root causes of degradation.
From an industrial chemistry standpoint, resistance to oxidation depends on molecular structure, impurity profile, packaging compatibility, and manufacturing controls. At SANDOO PHARMACEUTICALS AND CHEMICALS CO.,LTD., we design oxidative-resistant systems by optimizing excipient ratios, refining purification steps, and implementing strict oxygen control during blending and filling.
How Do Molecular Structure and Antioxidant Systems Improve Stability?
Certain Formulations demonstrate superior oxidative resistance because of deliberate molecular engineering and functional excipient selection. The presence of electron-withdrawing groups, steric hindrance, and stable ring systems can significantly reduce susceptibility to free radical attack. Additionally, chelating agents bind trace metals, preventing catalytic oxidation reactions.
An effective antioxidant system typically includes primary antioxidants that donate hydrogen atoms, secondary antioxidants that decompose peroxides, and synergists that regenerate the active antioxidant species. Our development team carefully evaluates compatibility through forced degradation studies and stress profiling.
The following table summarizes core technical parameters that influence oxidative stability in high-performance Formulations developed in our facility.
| Parameter | Standard Range | Impact on Oxidative Stability |
| Oxygen Residual Level | Below 1.5% | Reduces radical initiation rate |
| Moisture Content | Below 0.5% | Limits hydrolytic and oxidative synergy |
| Antioxidant Concentration | 0.05% to 0.2% | Prevents peroxide propagation |
| Metal Ion Control | Below 10 ppm | Minimizes catalytic oxidation |
| pH Stability Window | 5.0 to 7.0 | Maintains molecular integrity |
Through validated analytical tools such as HPLC assay, peroxide value testing, and GC residual solvent analysis, our quality control department ensures each batch meets strict stability specifications. This structured approach enables our factory to maintain consistent oxidative resistance across production cycles.
What Role Do Manufacturing Environment and Packaging Play?
Even the most robust chemical design may fail if environmental controls are inadequate. Controlled atmosphere blending, nitrogen blanketing, and low-humidity cleanrooms are essential in preventing premature oxidation. In pharmaceutical operations, oxygen-sensitive APIs require sealed transfer systems and minimized headspace exposure.
Packaging materials also determine long-term stability. High-barrier aluminum blisters, multilayer laminated pouches, and UV-protective containers reduce oxygen diffusion and photodegradation. Our packaging validation includes oxygen transmission rate testing and compatibility analysis to ensure product integrity throughout the declared shelf life.
The next table highlights packaging-related performance indicators commonly applied in oxidative-resistant Formulations manufactured by SANDOO PHARMACEUTICALS AND CHEMICALS CO.,LTD.
| Packaging Factor | Technical Specification | Stability Contribution |
| Oxygen Transmission Rate | Below 5 cc per m2 per day | Prevents external oxygen penetration |
| Light Protection | UV filtration above 99% | Reduces photochemical oxidation |
| Headspace Control | Nitrogen flushing | Limits internal oxidation trigger |
| Seal Integrity | Zero leakage at 30 kPa | Maintains barrier performance |
By integrating environmental engineering with validated packaging systems, our production lines consistently deliver stable Formulations suitable for global distribution channels.
How Do Quality Control and Regulatory Standards Strengthen Degradation Resistance?
In highly regulated markets, oxidative resistance must be proven through documented stability protocols. Accelerated testing at 40 degrees Celsius and 75 percent relative humidity simulates long-term degradation patterns. Real-time studies confirm shelf-life projections under labeled storage conditions.
Our factory implements GMP-compliant batch documentation, in-process oxygen monitoring, and impurity profiling. Risk assessments identify potential degradation pathways before commercial scale-up. These preventive controls reduce variability and ensure reproducible stability performance.
At SANDOO, we also conduct forced oxidation studies using peroxide stress and photostability testing to predict degradation behavior. The data obtained guide formulation refinement and packaging optimization. Through this systematic approach, our solutions consistently achieve extended shelf life and reliable potency retention.
Why Do Certain Formulations Maintain Performance in Harsh Storage Conditions?
Products distributed across North America, Southeast Asia, and the Middle East face temperature fluctuations and high humidity during transportation. Stability under these conditions depends on a balanced combination of molecular resilience, antioxidant protection, environmental control, and protective packaging.
Our development philosophy emphasizes real-world simulation. Shipping stress studies, vibration testing, and climatic chamber validation ensure that finished Formulations maintain assay compliance, color stability, and functional performance. These measures protect supply chain integrity and safeguard customer confidence.
By aligning chemical engineering, material science, and regulatory expertise, our factory provides dependable solutions tailored to demanding industrial and pharmaceutical markets. This integrated model allows our teams to respond quickly to custom specifications while maintaining strict quality benchmarks.
Frequently Asked Questions
Q1: What Makes Certain Formulations More Resistant to Oxidation and Degradation?
Resistance is achieved through optimized molecular design, effective antioxidant systems, strict control of oxygen and moisture, validated packaging barriers, and comprehensive stability testing. Each factor works together to prevent radical formation and slow degradation kinetics under both accelerated and long-term storage conditions.
Q2: How can manufacturers verify oxidative stability before commercial launch?
Manufacturers perform forced degradation studies, accelerated climatic testing, impurity profiling, and oxygen transmission analysis. These evaluations reveal potential weaknesses and allow reformulation before scale-up, ensuring regulatory compliance and consistent shelf life.
Q3: Why does packaging selection significantly influence long-term stability?
Packaging determines oxygen exposure, light protection, and moisture ingress. High-barrier materials combined with nitrogen flushing and seal validation prevent environmental triggers that accelerate oxidation, thereby preserving potency and product appearance throughout distribution.
In competitive pharmaceutical and specialty chemical markets, oxidative resistance directly impacts safety, compliance, and brand reputation. Through controlled manufacturing, advanced analytical testing, and optimized antioxidant strategies, SANDOO PHARMACEUTICALS AND CHEMICALS CO.,LTD. delivers reliable and durable solutions tailored to global standards. If you are seeking stable, high-performance systems designed for extended shelf life and demanding environments, contact our technical team today to discuss your project requirements and request detailed specifications.